
However, studies that have stratified LAM outcomes by CD4 count are limited, and there are no published data on CD4-stratified LAM-related outcomes in unselected TB suspects from a primary care setting. Recent studies have shown that urine LAM may have diagnostic value in HIV-infected, ,, but not in HIV-uninfected TB suspects. cerebro-spinal fluid and pleural fluid, and a user-friendly dipstick prototype of the test has been developed and is currently being validated as a POC test. Moreover, the assay has potential to be applied to other biological fluids e.g. This test option is attractive because urine is a sterile and easily obtainable biological fluid that can be assayed even in sputum scarce patients. More recently a standardised and commercially available lipoarabinomannan (LAM) antigen-detection assay has been developed (Clearview® TB ELISA, Inverness Medical Innovations, USA see ), which now supersedes a pre-commercial prototype (MTB LAM ELISA Test®, Chemogen, Portland, USA) first tested in 2005 in Tanzania, and based on earlier developmental studies. The search for a rapid point-of-care (POC) test continues. Newer technologies such as the T cell assays are not useful as rule-in tests for the diagnosis of active TB in adults, and molecular assays are not widely available in high burden countries. Mycobacterial culture results are only available after several weeks, if at all, in resource-poor settings. These drawbacks are exacerbated in HIV-infected patients, where the diagnostic ‘gap’ is the widest - smear-positivity in this sub-group is as low as 20%, and the clinical and the radiological features are often atypical. Delayed diagnosis facilitates disease transmission, increases healthcare costs, increases mortality, and causes greater lung damage resulting in chronic disability. Current tools for the diagnosis of TB are suboptimal and this contributes to poor control. Tuberculosis (TB) kills almost two million people annually and is out of control in Sub-Saharan Africa where up to 80% of TB patients may be co-infected with HIV. 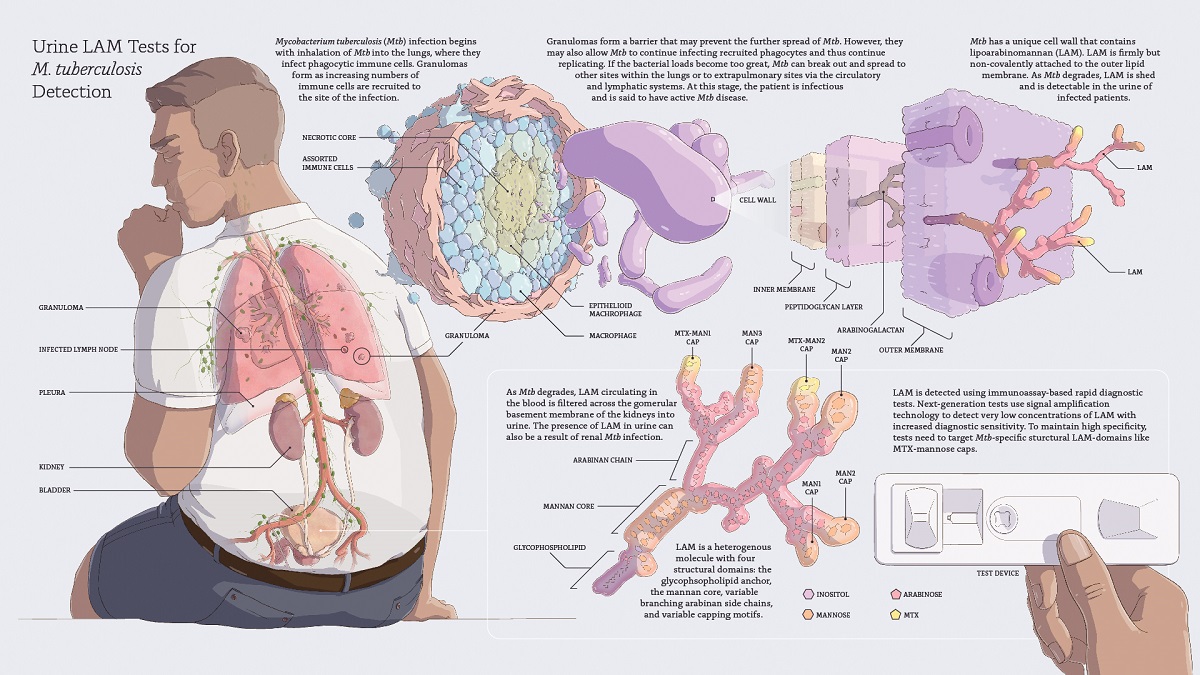 
The funders had no role in study design, data collection and analysis, decision to publish, or preparation of the manuscript.Ĭompeting interests: The authors have declared that no competing interests exist. is also supported by a Medical Research Council (MRC) Career Development Award and a NRF/SARChI award, and M.P. and A.Z.), UK Cambridge Biomedical Research Centre (CBRC), National Institute for Health Research (NIHR) (A.Z.), and in by part by the Canadian Institutes of Health Research (CIHR) grant MOP-89918 M.P., K.D.). This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.įunding: This study was supported by a TBsusgent grant from the European Commission (EU-FP7 K.D., M.P., M.H.), Europe AID ADAT grant (A.Z. Received: JanuAccepted: MaPublished: March 24, 2010Ĭopyright: © 2010 Dheda et al. PLoS ONE 5(3):Įditor: Ben Marais, University of Stellenbosch, South Africa (2010) Clinical Utility of a Commercial LAM-ELISA Assay for TB Diagnosis in HIV-Infected Patients Using Urine and Sputum Samples. Citation: Dheda K, Davids V, Lenders L, Roberts T, Meldau R, Ling D, et al.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
